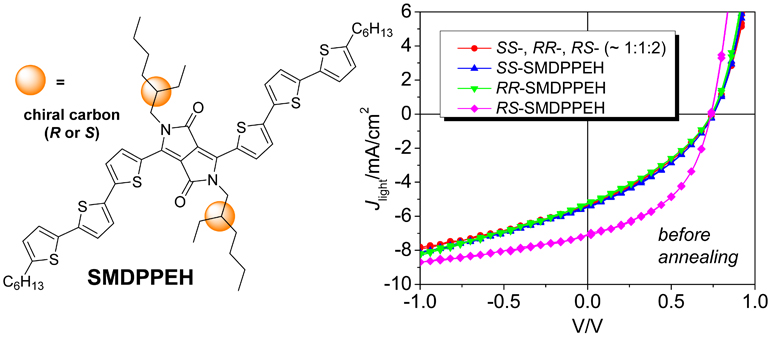
Three stereochemically pure isomers and two isomeric mixtures of a solution-processable diketopyrrolopyrrole-containing oligothiophene (SMDPPEH) have been used to study the effect of 2-ethylhexyl solubilizing group stereochemistry on the film morphology and bulk heterojunction (BHJ) solar cell characteristics of small molecule organic photovoltaics. The different SMDPPEH stereoisomer compositions exhibit nearly identical optoelectronic properties in the molecularly dissolved state, as well as in amorphous films blended with PCBM. However, for films in which SMDPPEH crystallization is induced by thermal annealing, significant differences in molecular packing between the different stereoisomer formulations are observed. These differences are borne out in photovoltaic device characteristics for which unannealed devices show very similar behavior, while after annealing the RR– and SS-SMDPPEH enantiomers show blue-shifted peak EQE relative to the SMDPPEH isomer mixtures. Unannealed devices made from the most crystalline stereoisomer, meso RS-SMDPPEH, are not completely amorphous, and show improved photocurrent generation as a result. Unlike the other compounds, after thermal annealing the RS-SMDPPEH devices show reduced device performance. The results reveal that the chirality of commonly used 2-ethylhexyl solubilizing chains can have a significant effect on the morphology, absorption, and optimum processing conditions of small molecule organic thin films used as photovoltaic device active layers.
