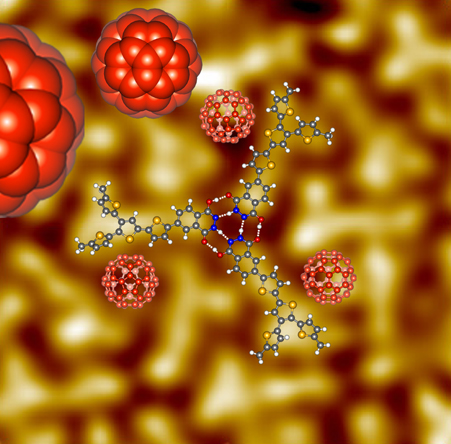
We previously reported that a branched quaterthiophene donor chromophore functionalized with a phthalhydrazide hydrogen bonding (H-bonding) unit (MeBQPH) gives two-fold more efficient bulk heterojunction organic solar cells (with C60 acceptors) compared to a nearly identical donor incapable of H-bonding (MeBQPME). Here, scanning tunneling microscopy (STM) studies confirm the formation of MeBQPH trimer rosettes on Au(111) through phthalhydrazide H-bonding interactions that are sufficiently strong to compete with adsorbate/substrate interactions. The MeBQPME comparator molecule void of hydrogen bonding functionality does not similarly assemble on the metal surface. Complementary density functional theory (DFT) calculations facilitate a structural understanding of the MeBQPH donor assemblies and their strong stabilization through formation of six hydrogen bonds. STM studies also reveal the templated growth of C60 on ordered MeBQPH monolayers with C60 molecules preferentially occupying the three-fold interstitial sites of the MeBQPH monolayer. This work supports the idea that H-bonding interactions can be used to control the morphology of organic donor–acceptor blends to potentially create efficient and stable bulk heterojunction photovoltaic devices.
